 (k) The subatomic particle not present in a hydrogen atom is. (j) The maximum number of electrons that can go into the M shell is. (i) The maximum number of electrons that can be accommodated in L shell are. 
(h) The anode rays obtained from hydrogen gas consist of particles called. A: Yes, it can happen but very, very infrequently. (g) Cathode rays are a beam of fast-moving. Q: If a neutron decays into a proton and an electron does it form a hydrogen atom as an end product. (f) Almost all the mass of an atom is concentrated in a small region of space called the. (e) If the nucleus of an atom has atomic number 17, mass number 37 and there are 17 electrons outside the nucleus, the number of neutrons in it is. electrons.(d) An atom of an element has 11 protons, 11 electrons and 12 neutrons. (c) An atom has atomic mass number 23 and atomic number 11. (b) The total number of protons and neutrons in the nucleus of an atom is called its. Fill in the blanks in the following statements :(a) The number of protons in the nucleus of an atom is called its.If an atom contains one electron and one proton, will it carry any charge or not?. 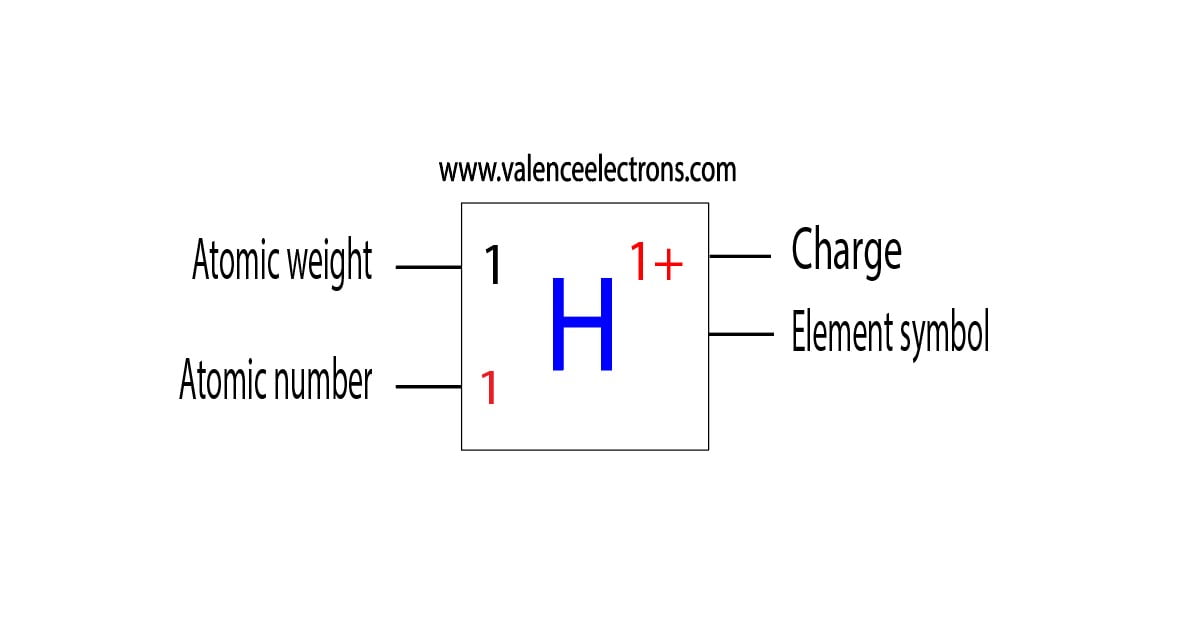
Therefore,it is neutral.(c) The mass of an electron is about 1/2000 times that of proton.(d) An isotope of iodine is used for making tinctures of iodine, which is used as a medicine. Thomson proposed that the nucleus of an atom contains only nucleons.(b) A neutron is formed by an electron and a proton combining together. For the following statements, write T for True and F for False.(a) J.J.Compare an electron, a proton and a neutron in respect of their relative masses and charges.Is it possible for the atom of an element to have one electron, one proton and no neutron.Goldstein's experiments which involved passing high voltage electricity through gases at very low pressure resulted in the discovery of :(a) electron (b) proton (c) nucleus (d) neutron.On the basis of Rutherford’s model of an atom, which sub-atomic particle is present in the nucleus of an atom?.Which of the following statement is correct about the atom of an element?(a) an atom can have only protons and neutrons but no electrons.(b) an atom can have only electrons and neutrons but no protons(c) an atom can have only electron and proton but no neutron(d) an atom must always have a proton, neutron and electron.What is the usual symbol for (a) an electron (b) a proton, and (c) a neutron?.The isotope deuterium of hydrogen has(a) no neutrons and one proton (b) one neutron and two protons(c) one electron and two neutron (d) one proton and one neutron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
